| Citation: | Ye H G, Su Z C, Tang F, Bao Y T, Lao X Z et al. Probing defects in ZnO by persistent phosphorescence. Opto-Electron Adv 1, 180011 (2018). doi: 10.29026/oea.2018.180011 |
-
Abstract
Native point defects in ZnO are so complicated that most of them are still debating issues, although they have been studied for decades. In this paper, we experimentally reveal two sub-components usually hidden in the low energy tail of the main broad green luminescence band peaking at 547 nm (~2.267 eV) in intentionally undoped ZnO single crystal by selecting the below-band-gap (BBG) optical excitations (e.g. light wavelengths of 385 nm and 450 nm). Moreover, both sub-components are manifested as long persistent phosphorescence once the BBG excitations are removed. With the aid of a newly developed model, the energy depths of two electron traps involved within the long lived orange luminescence are determined to be 44 meV and 300 meV, respectively. The candidates of these two electron traps are argued to be most likely hydrogen and zinc interstitials in ZnO.-
Keywords:
- zinc oxide /
- defects /
- phosphorescence /
- photoluminescence
-
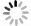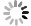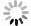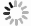
-
References
[1] Hirschwald W H. Zinc oxide: an outstanding example of a binary compound semiconductor. Acc Chem Res 18, 228-234 (1985). doi: 10.1021/ar00116a001 [2] Jagadish C, Pearton S J. Zinc Oxide Bulk, Thin Films and Nanostructures: Processing, Properties, and Applications (Elsevier, Amsterdam, 2006). [3] Klingshirn C F, Waag A, Hoffmann A, Geurts J. Zinc Oxide: From Fundamental Properties Towards Novel Applications (Springer, Berlin Heidelberg, 2010). [4] Sun X W, Yi Y. ZnO Nanostructures and Their Applications (CRC Press, New York, 2016). [5] Özgür Ü, Alivov Y I, Liu C, Teke A, Reshchikov M A et al. A comprehensive review of ZnO materials and devices. J Appl Phys 98, 041301 (2005). doi: 10.1063/1.1992666 [6] Srikant V, Clarke D R. On the optical band gap of zinc oxide. J Appl Phys 83, 5447-5451 (1998). doi: 10.1063/1.367375 [7] Kohan A F, Ceder G, Morgan D, Van de Walle C G. First-principles study of native point defects in ZnO. Phys Rev B 61, 15019-15027 (2000). doi: 10.1103/PhysRevB.61.15019 [8] Zhang S B, Wei S H, Zunger A. Intrinsic n-type versus p-type doping asymmetry and the defect physics of ZnO. Phys Rev B 63, 075205 (2001). doi: 10.1103/PhysRevB.63.075205 [9] Shi S L, Li G Q, Xu S J, Zhao Y, Chen G H. Green luminescence band in ZnO: fine structures, electron-phonon coupling, and temperature effect. J Phys Chem B 110, 10475-10478 (2006). doi: 10.1021/jp0610968 [10] Sokol A A, French S A, Bromley S T, Catlow R A, Van Dam H J et al. Point defects in ZnO. Faraday Discuss 134, 267-282 (2007). doi: 10.1039/B607406E [11] Janotti A, Van de Walle C G. Native point defects in ZnO. Phys Rev B 76, 165202 (2007). doi: 10.1103/PhysRevB.76.165202 [12] McCluskey M D, Jokela S J. Defects in ZnO. J Appl Phys 106, 071101 (2009). doi: 10.1063/1.3216464 [13] Dai X M, Xu S J, Ling C C, Brauer G, Anwand W et al. Emission bands of nitrogen-implantation induced luminescent centers in ZnO crystals: experiment and theory. J Appl Phys 112, 046102 (2012). doi: 10.1063/1.4749402 [14] Fan J C, Sreekanth K M, Xie Z, Chang S L, Rao K V. P-type ZnO materials: theory, growth, properties and devices. Prog Mater Sci 58, 874-985 (2013). doi: 10.1016/j.pmatsci.2013.03.002 [15] Bollmann J, Simon D K. Deep level defects in ZnO. Physica B Condens Matter 439, 14-19 (2014). doi: 10.1016/j.physb.2013.11.006 [16] Chen Y N, Xu S J, Zheng C C, Ning J Q, Ling F C C et al. Nature of red luminescence band in research-grade ZnO single crystals: a "self-activated" configurational transition. Appl Phys Lett 105, 041912 (2014). doi: 10.1063/1.4892356 [17] Ding L, Li B K, He H T, Ge W K, Wang J N et al. Classification of bound exciton complexes in bulk ZnO by magnetophotoluminescence spectroscopy. J Appl Phys 105, 053511 (2009). doi: 10.1063/1.3087762 [18] Chen Y N, Zheng C C, Ning J Q, Wang R X, Ling C C et al. Who make transparent ZnO colorful?-Ion implantation and thermal annealing effects. Superlattices Microstruct 99, 208-213 (2016). doi: 10.1016/j.spmi.2016.02.022 [19] Ye H G, Su Z C, Tang F, Wang M Z, Chen G D et al. Excitation dependent phosphorous property and new model of the structured green luminescence in ZnO. Sci Rep 7, 41460 (2017). doi: 10.1038/srep41460 [20] Alkauskas A, Pasquarello A. Band-edge problem in the theoretical determination of defect energy levels: the O vacancy in ZnO as a benchmark case. Phys Rev B 84, 125206 (2011). doi: 10.1103/PhysRevB.84.125206 [21] Koßmann J, Hättig C. Investigation of interstitial hydrogen and related defects in ZnO. Phys Chem Chem Phys 14, 16392-16399 (2012). doi: 10.1039/c2cp42928d [22] Lyons J L, Alkauskas A, Janotti A, Van de Walle C G. Deep donor state of the copper acceptor as a source of green luminescence in ZnO. Appl Phys Lett 111, 042101 (2017). doi: 10.1063/1.4995404 [23] Ye H G, Su Z C, Tang F, Chen G D, Wang J et al. Role of free electrons in phosphorescence in n-type wide bandgap semiconductors. Phys Chem Chem Phys 19, 30332-30338 (2017). doi: 10.1039/C7CP05796B [24] Rodnyi P A, Khodyuk I V. Optical and luminescence properties of zinc oxide (Review). Opt Spectrosc 111, 776-785 (2011). doi: 10.1134/S0030400X11120216 [25] Wang X H, Xu S J. Two-electron-satellite transition of donor bound exciton in ZnO: radiative Auger effect. Appl Phys Lett 102, 181909 (2013). doi: 10.1063/1.4804619 [26] Xu S J, Xiong S J, Shi S L. Resonant coupling of bound excitons with LO phonons in ZnO: excitonic polaron states and Fano interference. J Chem Phys 123, 221105 (2005). doi: 10.1063/1.2140701 [27] Heitz R, Hoffmann A, Broser I. Fe3+ center in ZnO. Phys Rev B 45, 8977-8988 (1992). [28] Melnick D A. Zinc oxide photoconduction, an oxygen adsorption process. J Chem Phys 26, 1136-1146 (1957). doi: 10.1063/1.1743483 [29] Takahashi Y, Kanamori M, Kondoh A, Minoura H, Ohya Y. Photoconductivity of ultrathin zinc oxide Films. Jpn J Appl Phys 33, 6611-6615 (1994). doi: 10.1143/JJAP.33.6611 [30] Murphy T E, Moazzami K, Phillips J D. Trap-related photoconductivity in ZnO epilayers. J Electron Mater 35, 543-549 (2006). doi: 10.1007/s11664-006-0097-x [31] Liao Z M, Lu Y, Xu J, Zhang J M, Yu D P. Temperature dependence of photoconductivity and persistent photoconductivity of single ZnO nanowires. Appl Phys A 95, 363-366 (2009). doi: 10.1007/s00339-008-5058-1 [32] Lany S, Zunger A. Anion vacancies as a source of persistent photoconductivity in Ⅱ-Ⅵ and chalcopyrite semiconductors. Phys Rev B 72, 035215 (2005). doi: 10.1103/PhysRevB.72.035215 [33] Kang Y, Nahm H H, Han S. Light-induced peroxide formation in ZnO: origin of persistent photoconductivity. Sci Rep 6, 35148 (2016). doi: 10.1038/srep35148 [34] Jeffries B T, Gonzalez R, Chen Y, Summers G P. Luminescence in thermochemically reduced MgO: the role of hydrogen. Phys Rev B 25, 2077-2080 (1982). doi: 10.1103/PhysRevB.25.2077 [35] Li Y, Gecevicius M, Qiu J R. Long persistent phosphors—from fundamentals to applications. Chem Soc Rev 45, 2090-2136 (2016). doi: 10.1039/C5CS00582E [36] Janotti A, Van de Walle C G. Fundamentals of zinc oxide as a semiconductor. Rep Prog Phys 72, 126501 (2009). doi: 10.1088/0034-4885/72/12/126501 [37] Oba F, Choi M, Togo A, Tanaka I. Point defects in ZnO: an approach from first principles. Sci Technol Adv Mater 12, 034302 (2011). doi: 10.1088/1468-6996/12/3/034302 [38] Van de Walle C G. Hydrogen as a cause of doping in Zinc Oxide. Phys Rev Lett 85, 1012-1015 (2000). doi: 10.1103/PhysRevLett.85.1012 [39] Hofmann D M, Hofstaetter A, Leiter F, Zhou H J, Henecker F et al. Hydrogen: a relevant shallow donor in Zinc Oxide. Phys Rev Lett 88, 045504 (2002). doi: 10.1103/PhysRevLett.88.045504 [40] Qiu H S, Meyer B, Wang Y M, Wöll C. Ionization energies of shallow donor states in ZnO created by reversible formation and depletion of H interstitials. Phys Rev Lett 101, 236401 (2008). doi: 10.1103/PhysRevLett.101.236401 [41] Dingle R. Luminescent transitions associated with divalent copper impurities and the green emission from semiconducting Zinc Oxide. Phys Rev Lett 23, 579-581 (1969). doi: 10.1103/PhysRevLett.23.579 [42] Garces N Y, Wang L, Bai L, Giles N C, Halliburton L E. Role of copper in the green luminescence from ZnO crystals. Appl Phys Lett 81, 622-624 (2002). doi: 10.1063/1.1494125 [43] Ye J D, Gu S L, Qin F, Zhu S M, Liu S M et al. Correlation between green luminescence and morphology evolution of ZnO films. Appl Phys A 81, 759-762 (2005). doi: 10.1007/s00339-004-2996-0 [44] Čížek J, Valenta J, Hruška P, Melikhova O, Procházka I et al. Origin of green luminescence in hydrothermally grown ZnO single crystals. Appl Phys Lett 106, 251902 (2015). doi: 10.1063/1.4922944 [45] Prucnal S, Wu J D, Berencén Y, Liedke M O, Wagner A et al. Engineering of optical and electrical properties of ZnO by non-equilibrium thermal processing: the role of zinc interstitials and zinc vacancies. J Appl Phys 122, 035303 (2017). doi: 10.1063/1.4994796 [46] Zhou D M, Kittilstved K R. Electron trapping on Fe3+ sites in photodoped ZnO colloidal nanocrystals. Chem Commun 52, 9101-9104 (2016). doi: 10.1039/C6CC00514D [47] Chen T X, Cao L, Zhang W H, Zhang W, Han Y Y et al. Correlation between electronic structure and magnetic properties of Fe-doped ZnO films. J Appl Phys 111, 123715 (2012). doi: 10.1063/1.4730605 -
Access History
Article Metrics
-
Figure 1.
(Colour on-line) PL spectra of the ZnO bulk crystal at 10 K.
-
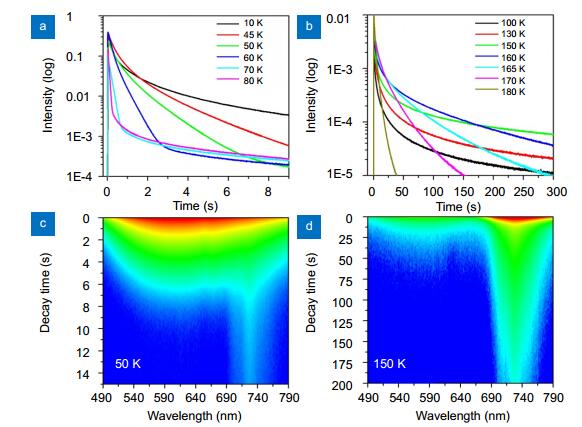
Figure 2.
(a, b) (Colour on-line) Decaying traces of the visible luminescence of the sample at different temperatures after ceasing the 385 nm excitation. (c, d) show the recorded time-resolved phosphorescence images at 50 and 150 K, respectively.
-
Figure 3.
(Colour on-line) Variable-temperature PL spectra of the sample under the continuous excitation of 450 nm laser.

 E-mail Alert
E-mail Alert RSS
RSS


 DownLoad:
DownLoad: