| Citation: | Lee E, Sun B, Luo J Q, Singh S, Choudhury D et al. Compact pulsed thulium-doped fiber laser for topographical patterning of hydrogels. Opto-Electron Adv 3, 190039 (2020). doi: 10.29026/oea.2020.190039 |
Compact pulsed thulium-doped fiber laser for topographical patterning of hydrogels
-
Abstract
We report the generation of high energy 2 μm picosecond pulses from a thulium-doped fiber master oscillator power amplifier system. The all-fiber configuration was realized by a flexible large-mode area photonic crystal fiber (LMA-PCF). The amplifier output is a linearly-polarized 1.5 ns, 100 kHz pulse train with a pulse energy of up to 250 μJ. Pulse compression was achieved with (2+2)-pass chirped volume Bragg grating (CVBG) to obtain a 2.8 ps pulse width with a total pulse energy of 46 μJ. The overall system compactness was enabled by the all-fiber amplifier design and the multi-pass CVBG-based compressor. The laser output was then used to demonstrate high-speed direct-writing capability on a temperature-sensitive biomaterial to change its topography (i.e. fabricate microchannels, foams and pores). The topographical modifications of biomaterials are known to influence cell behavior and fate which is potentially useful in many cell and tissue engineering applications. -
-
References
[1] Gehlich N, Bonhoff T, Sisken L, Ramme M, Gaida C et al. Utilizing the transparency of semiconductors via "backside" machining with a nanosecond 2 μm Tm:fiber laser. Proc SPIE 8968, 89680W (2014). doi: 10.1117/12.2040306 [2] Voisiat B, Gaponov D, Gečys P, Lavoute L, Silva M et al. Material processing with ultra-short pulse lasers working in 2μm wavelength range. Proc SPIE 9350, 935014 (2015). doi: 10.1117/12.2078651 [3] Blackmon R L, Fried N M, Irby P B. Comparison of holmium:YAG and thulium fiber laser lithotripsy: ablation thresholds, ablation rates, and retropulsion effects. J Biomed Opt 16, 071403 (2011). doi: 10.1117/1.3564884 [4] Jansen F, Stutzki F, Jauregui C, Limpert J, Tünnermann A. High-power very large mode-area thulium-doped fiber laser. Opt Lett 37, 4546-4548 (2012). doi: 10.1364/OL.37.004546 [5] Maine P, Strickland D, Bado P, Pessot M, Mourou G. Generation of ultrahigh peak power pulses by chirped pulse amplification. IEEE J Quantum Electron 24, 398-403 (1988). doi: 10.1109/3.137 [6] Gaida C, Gebhardt M, Heuermann T, Stutzki F, Jauregui C et al. Ultrafast thulium fiber laser system emitting more than 1 kW of average power. Opt Lett 43, 5853-5856 (2018). doi: 10.1364/OL.43.005853 [7] Gaida C, Gebhardt M, Stutzki F, Jauregui C, Limpert J et al. 90 fs pulses with > 5 GW peak power from a high repetition rate Tm-doped fiber CPA system. In Advanced Solid State Lasers 2017 (Optical Society of America, 2017);https://doi.org/10.1364/ASSL.2017.ATh3A.5. [8] Gaponov D, Lavoute L, Ducros N, Hideur A, Février S. 10 μJ-Class compact thulium all-fibered CPA system. In 2017 Conference on Lasers and Electro-Optics Europe and European Quantum Electronics Conference (Optical Society of America, 2017). [9] Luo J Q, Sun B, Ji J H, Tan E L, Zhang Y et al. High-efficiency femtosecond Raman soliton generation with a tunable wavelength beyond 2μm. Opt Lett 42, 1568-1571 (2017). doi: 10.1364/OL.42.001568 [10] Lyot B. Optical apparatus with wide field using interference of polarized light. C R Acad Sci 197, 1593 (1933). doi: 10.1007/s12596-015-0256-7 [11] Imani R, Emami S H, Moshtagh P R, Baheiraei N, Sharifi A M. Preparation and characterization of agarose-gelatin blend hydrogels as a cell encapsulation matrix: an in-vitro study. J Macromol Sci, Part B 51, 1606-1616 (2012). doi: 10.1080/00222348.2012.657110 [12] Sincore A, Bradford J D, Cook J, Shah L, Richardson M C. High average power thulium-doped silica fiber lasers: review of systems and concepts. IEEE J Sel Top Quantum Electron 24, 0901808 (2018). doi: 10.1109/JSTQE.2017.2775964 [13] Schimpf D N, Seise E, Limpert J, Tünnermann A. Decrease of pulse-contrast in nonlinear chirped-pulse amplification systems due to high-frequency spectral phase ripples. Opt Express 16, 8876-8886 (2008). doi: 10.1364/OE.16.008876 [14] Schimpf D N, Seise E, Limpert J, Tünnermann A. Self-phase modulation compensated by positive dispersion in chirped-pulse systems. Opt Express 17, 4997-5007 (2009). doi: 10.1364/OE.17.004997 [15] Lee E, Luo J Q, Sun B, Ramalingam V, Zhang Y et al. Flexible single-mode delivery of a high-power 2μm pulsed laser using an antiresonant hollow-core fiber. Opt Lett 43, 2732-2735 (2018). doi: 10.1364/OL.43.002732 [16] Schimpf D N, Limpert J, Tünnermann A. Controlling the influence of SPM in fiber-based chirped-pulse amplification systems by using an actively shaped parabolic spectrum. Opt Express 15, 16945-16953 (2007). doi: 10.1364/OE.15.016945 [17] Chen Y H, Raghuraman S, Ho D, Tang D Y, Yoo S. Normal dispersion thulium fiber for ultrafast near-2 μm fiber laser. In 2018 Conference on Lasers and Electro-Optics: CLEO: Applications and Technology 2018 (Optical Society of America, 2018); https://doi.org/10.1364/CLEO_AT.2018.AM2M.2. [18] Bartulevicius T, Frankinas S, Michailovas A, Vasilyeu R, Smirnov V et al. Compact fiber CPA system based on a CFBG stretcher and CVBG compressor with matched dispersion profile. Opt Express 25, 19856-19862 (2017). doi: 10.1364/OE.25.019856 [19] Turunen S, Haaparanta A M, Äänismaa R, Kellomäki M. Chemical and topographical patterning of hydrogels for neural cell guidance in vitro. J Tissue Eng Regen Med 7, 253-270 (2013). doi: 10.1002/term.520 [20] Yu T Y, Ober C K. Methods for the topographical patterning and patterned surface modification of hydrogels based on hydroxyethyl methacrylate. Biomacromolecules 4, 1126-1131 (2003). doi: 10.1021/bm034079m [21] Nikkhah M, Edalat F, Manoucheri S, Khademhosseini A. Engineering microscale topographies to control the cell-substrate interface. Biomaterials 33, 5230-5246 (2012). doi: 10.1016/j.biomaterials.2012.03.079 [22] Kim H N, Jiao A, Hwang N S, Kim M S, Kang D H et al. Nanotopography-guided tissue engineering and regenerative medicine. Adv Drug Deliv Rev 65, 536-558 (2013). doi: 10.1016/j.addr.2012.07.014 [23] Dalby M J, Gadegaard N, Oreffo R O C. Harnessing nanotopography and integrin-matrix interactions to influence stem cell fate. Nat Mater 13, 558-569 (2014). doi: 10.1038/nmat3980 [24] Brandl F, Sommer F, Goepferich A. Rational design of hydrogels for tissue engineering: impact of physical factors on cell behavior. Biomaterials 28, 134-146 (2007). doi: 10.1016/j.biomaterials.2006.09.017 [25] Burg K J, Porter S, Kellam J F. Biomaterial developments for bone tissue engineering. Biomaterials 21, 2347-2359 (2000). doi: 10.1016/S0142-9612(00)00102-2 [26] Ranella A, Barberoglou M, Bakogianni S, Fotakis C, Stratakis E. Tuning cell adhesion by controlling the roughness and wettability of 3D micro/nano silicon structures. Acta Biomater 6, 2711-2720 (2010). doi: 10.1016/j.actbio.2010.01.016 [27] Anene-Nzelu C G, Choudhury D, Li H P, Fraiszudeen A, Peh K Y et al. Scalable cell alignment on optical media substrates. Biomaterials 34, 5078-5087 (2013). doi: 10.1016/j.biomaterials.2013.03.070 [28] Falconnet D, Csucs G, Grandin H M, Textor M. Surface engineering approaches to micropattern surfaces for cell-based assays. Biomaterials 27, 3044-3063 (2006). doi: 10.1016/j.biomaterials.2005.12.024 [29] Chaudhari A A, Vig K, Baganizi D R, Sahu R, Dixit S et al. Future prospects for scaffolding methods and biomaterials in skin tissue engineering: a review. Int J Mol Sci 17, 1974 (2016). doi: 10.3390/ijms17121974 [30] Liu X H, Ma P X. Polymeric scaffolds for bone tissue engineering. Ann Biomed Eng 32, 477-486 (2004). doi: 10.1023/B:ABME.0000017544.36001.8e [31] Guillemot F, Souquet A, Catros S, Guillotin B, Lopez J et al. High-throughput laser printing of cells and biomaterials for tissue engineering. Acta Biomater 6, 2494-2500 (2010). doi: 10.1016/j.actbio.2009.09.029 [32] Subia B, Kundu J, Kundu S C. Biomaterial scaffold fabrication techniques for potential tissue engineering applications. Tissue Eng 141 (InTech. 2010). [33] Chrisey D B. The power of direct writing. Science 289, 879-881 (2000). doi: 10.1126/science.289.5481.879 [34] Hribar K C, Soman P, Warner J, Chung P, Chen S C. Light-assisted direct-write of 3D functional biomaterials. Lab Chip 14, 268-275 (2014). doi: 10.1039/C3LC50634G [35] Wu P K, Ringeisen B R, Krizman D B, Frondoza C G, Brooks M et al. Laser transfer of biomaterials: Matrix-assisted pulsed laser evaporation (MAPLE) and MAPLE Direct Write. Rev Sci Instrum 74, 2546-2557 (2003). doi: 10.1063/1.1544081 [36] Johnston-Banks F. Gelatine. In Food Gels 233-289 (Springer, 1990); https://doi.org/10.1007/978-94-009-0755-3_7. [37] Tijore A, Irvine S A, Sarig U, Mhaisalkar P, Baisane V et al. Contact guidance for cardiac tissue engineering using 3D bioprinted gelatin patterned hydrogel. Biofabrication 10, 025003 (2018). doi: 10.1088/1758-5090/aaa15d [38] Kobuszewska A, Tomecka E, Zukowski K, Jastrzebska E, Chudy M et al. Heart-on-a-Chip: an investigation of the influence of static and perfusion conditions on cardiac (H9C2) cell proliferation, morphology, and alignment. SLAS Technol: Transl Life Sci Innov 22, 536-546 (2017). doi: 10.1177/2472630317705610 [39] Korin N, Bransky A, Khoury M, Dinnar U, Levenberg S. Design of well and groove microchannel bioreactors for cell culture. Biotechnol Bioeng 102, 1222-1230 (2009). doi: 10.1002/bit.22153 [40] Gaspard S, Oujja M, Abrusci C, Catalina F, Lazare S et al. Laser induced foaming and chemical modifications of gelatine films. J Photochem Photobiol A: Chem 193, 187-192 (2008). doi: 10.1016/j.jphotochem.2007.06.024 [41] Lazare S, Tokarev V, Sionkowska A, Wiśniewski M. Surface foaming of collagen, chitosan and other biopolymer films by KrF excimer laser ablation in the photomechanical regime. Appl Phys A 81, 465-470 (2005). doi: 10.1007/s00339-005-3260-y [42] Simoni R C, Lemes G F, Fialho S, Gonçalves O H, Gozzo A M et al. Effect of drying method on mechanical, thermal and water absorption properties of enzymatically crosslinked gelatin hydrogels. An Acad Bras Ciênc 89, 745-755 (2017). doi: 10.1590/0001-3765201720160241 [43] Emoto H, Kambic H, Chen J F, Nosé Y. Characterization of rehydrated gelatin gels. Artif Organs 15, 29-34 (1991). doi: 10.1111/j.1525-1594.1991.tb00756.x [44] Viswanathan P, Ondeck M G, Chirasatitsin S, Ngamkham K, Reilly G C et al. 3D surface topology guides stem cell adhesion and differentiation. Biomaterials 52, 140-147 (2015). doi: 10.1016/j.biomaterials.2015.01.034 [45] O'Brien F J, Harley B A, Yannas I V, Gibson L J. The effect of pore size on cell adhesion in collagen-GAG scaffolds. Biomaterials 26, 433-441 (2005). doi: 10.1016/j.biomaterials.2004.02.052 [46] Eiselt P, Yeh J, Latvala R K, Shea L D, Mooney D J. Porous carriers for biomedical applications based on alginate hydrogels. Biomaterials 21, 1921-1927 (2000). doi: 10.1016/S0142-9612(00)00033-8 [47] Van Tienen T G, Heijkants Ralf G J C, Buma P, de Groot J H, Pennings A J et al. Tissue ingrowth and degradation of two biodegradable porous polymers with different porosities and pore sizes. Biomaterials 23, 1731-1738 (2002). doi: 10.1016/S0142-9612(01)00280-0 -
Access History
Article Metrics
-
Figure 1.
Schematic of the system with the microscopy image of the thulium-doped large-mode area photonic crystal fiber (TD-LMA-PCF) cross-section and the splice point between the TD-LMA-PCF to a polarization-maintaining 30/250 silica fiber.
-
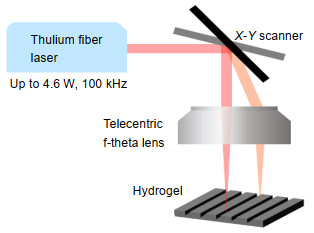
Figure 2.
Schematic of laser scanner setup for the topographical engineering of hydrogels.
-
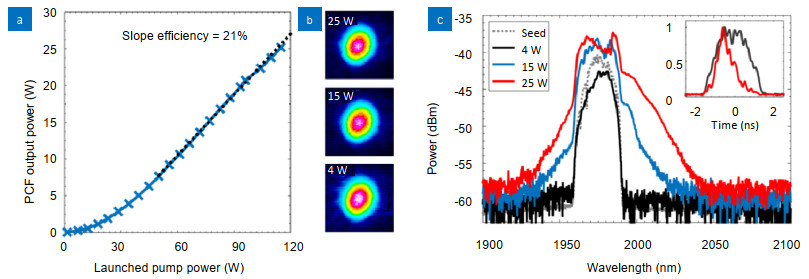
Figure 3.
(a) Slope efficiency curve of the main amplifier; (b) TD-LMA-PCF beam profile at different output powers; (c) Spectra of main amplifier seed (pre-amplifier 2 output) and main amplifier output at different pulse energies. Inset: Main amplifier output pulse measured using a fast photodetector.
-
Figure 4.
(a) Compressor efficiency and CVBG 1 leakage at different main amplifier output powers; (b) Autocorrelation trace of recompressed pulse at 1.7 W and 4.6 W average powers. Inset: Spectra at corresponding output powers after the compressor.
-
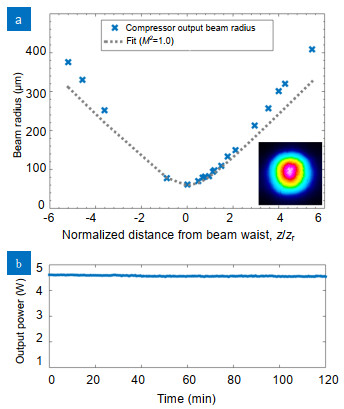
Figure 5.
(a) M2 measurement at the highest pulse energy after compressor. Inset: Beam profile at the highest pulse energy after compressor; (b) Power stability of the compressor output over 2 hours.
-
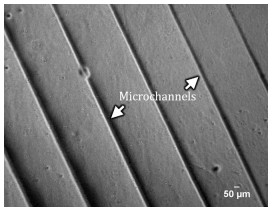
Figure 6.
Microscopy image of microchannels abricated on the hydrogel.
-
Figure 7.
(a) Schematic of laser scanner setup on dried hydrogel films. Inset: Schematic of foam formation due to water absorption within film causing laser-induced expansion. (b) Scanning electron microscopy images of foamed hydrogel films with decreasing photon flux (scanning speeds of 100, 120 and 150 mm/s at 4 W) from left to right.

 E-mail Alert
E-mail Alert RSS
RSS


 DownLoad:
DownLoad: