| Citation: | Jiang K, Liang SM, Sun XJ, Ben JW, Qu L et al. Rapid inactivation of human respiratory RNA viruses by deep ultraviolet irradiation from light-emitting diodes on a high-temperature-annealed AlN/Sapphire template. Opto-Electron Adv 6, 230004 (2023). doi: 10.29026/oea.2023.230004 |
Rapid inactivation of human respiratory RNA viruses by deep ultraviolet irradiation from light-emitting diodes on a high-temperature-annealed AlN/Sapphire template
-
Abstract
Efficient and eco-friendly disinfection of air-borne human respiratory RNA viruses is pursued in both public environment and portable usage. The AlGaN-based deep ultraviolet (DUV) light-emission diode (LED) has high practical potentials because of its advantages of variable wavelength, rapid sterilization, environmental protection, and miniaturization. Therefore, whether the emission wavelength has effects on the disinfection as well as whether the device is feasible to sterilize various respiratory RNA viruses under portable conditions is crucial. Here, we fabricate AlGaN-based DUV LEDs with different wavelength on high-temperature-annealed (HTA) AlN/Sapphire templates and investigate the inactivation effects for several respiratory RNA viruses. The AlN/AlGaN superlattices are employed between the template and upper n-AlGaN to release the strong compressive stress (SCS), improving the crystal quality and interface roughness. DUV LEDs with the wavelength of 256, 265, and 278 nm, corresponding to the light output power of 6.8, 9.6, and 12.5 mW, are realized, among which the 256 nm-LED shows the most potent inactivation effect in human respiratory RNA viruses, including SARS-CoV-2, influenza A virus (IAV), and human parainfluenza virus (HPIV), at a similar light power density (LPD) of ~0.8 mW/cm2 for 10 s. These results will contribute to the advanced DUV LED application of disinfecting viruses with high potency and broad spectrum in a portable and eco-friendly use.-
Keywords:
- AlGaN /
- DUV LED /
- superlattice /
- SARS-CoV-2 /
- influenza A virus
-
-
References
[1] Calderaro A, De Conto F, Buttrini M, Piccolo G, Montecchini S et al. Human respiratory viruses, including SARS-CoV-2, circulating in the winter season 2019–2020 in Parma, Northern Italy. Int J Infect Dis 102, 79–84 (2021). doi: 10.1016/j.ijid.2020.09.1473 [2] https://covid19.who.int. [3] https://www.who.int/publications/i/item/who-wer-9625-241-264. [4] Brankston G, Gitterman L, Hirji Z, Lemieux C, Gardam M. Transmission of influenza A in human beings. Lancet Infect Dis 7, 257–265 (2007). doi: 10.1016/S1473-3099(07)70029-4 [5] Duguid JP. The size and the duration of air-carriage of respiratory droplets and droplet-nuclei. Epidemiol Infect 44, 471–479 (1946). doi: 10.1017/S0022172400019288 [6] Liu Y, Ning Z, Chen Y, Guo M, Liu YL et al. Aerodynamic analysis of SARS-CoV-2 in two Wuhan hospitals. Nature 582, 557–560 (2020). doi: 10.1038/s41586-020-2271-3 [7] Cowling BJ, Ip DKM, Fang VJ, Suntarattiwong P, Olsen SJ et al. Aerosol transmission is an important mode of influenza A virus spread. Nat Commun 4, 1935 (2013). doi: 10.1038/ncomms2922 [8] Leung NHL. Transmissibility and transmission of respiratory viruses. Nat Rev Microbiol 19, 528–545 (2021). doi: 10.1038/s41579-021-00535-6 [9] Chin AWH, Lai AM Y, Peiris M, Poon LLM. SARS-CoV-2 Omicron variant is more stable than the ancestral strain on various surfaces. bioRxiv (2022).https://doi.org/10.1101/2022.03.09.483703 [10] Van Doremalen N, Bushmaker T, Morris DH, Holbrook MG, Gamble A et al. Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. N Engl J Med 382, 1564–1567 (2020). doi: 10.1056/NEJMc2004973 [11] Bean B, Moore BM, Sterner B, Peterson LR, Gerding ND et al. Survival of influenza viruses on environmental surfaces. J Infect Dis 146, 47–51 (1982). doi: 10.1093/infdis/146.1.47 [12] Zhou P, Yang XL, Wang XG, Hu B, Zhang L et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 579, 270–273 (2020). doi: 10.1038/s41586-020-2012-7 [13] Wise HM, Foeglein A, Sun JC, Dalton RM, Patel S et al. A complicated message: Identification of a novel PB1-related protein translated from influenza A virus segment 2 mRNA. J Virol 83, 8021–8031 (2009). doi: 10.1128/JVI.00826-09 [14] Eisfeld AJ, Neumann G, Kawaoka Y. At the centre: influenza A virus ribonucleoproteins. Nat Rev Microbiol 13, 28–41 (2015). doi: 10.1038/nrmicro3367 [15] Guerrero- Beltr·n JA, Barbosa- C·novas GV. Advantages and limitations on processing foods by UV light. Food Sci Technol Int 10, 137–147 (2004). doi: 10.1177/1082013204044359 [16] Nishisaka-Nonaka R, Mawatari K, Yamamoto T, Kojima M, Shimohata T et al. Irradiation by ultraviolet light-emitting diodes inactivates influenza a viruses by inhibiting replication and transcription of viral RNA in host cells. J Photochem Photobiol B 189, 193–200 (2018). doi: 10.1016/j.jphotobiol.2018.10.017 [17] Lo CW, Matsuura R, Iimura K, Wada S, Shinjo A et al. UVC disinfects SARS-CoV-2 by induction of viral genome damage without apparent effects on viral morphology and proteins. Sci Rep 11, 13804 (2021). doi: 10.1038/s41598-021-93231-7 [18] Bosshard F, Armand F, Hamelin R, Kohn T. Mechanisms of human adenovirus inactivation by sunlight and UVC light as examined by quantitative PCR and quantitative proteomics. Appl Environ Microbiol 79, 1325–1332 (2013). doi: 10.1128/AEM.03457-12 [19] Sarigiannis DA, Karakitsios SP, Antonakopoulou MP, Gotti A. Exposure analysis of accidental release of mercury from compact fluorescent lamps (CFLs). Sci Total Environ 435-436, 306–315 (2012). doi: 10.1016/j.scitotenv.2012.07.026 [20] Nunayon SS, Zhang HH, Lai ACK. Comparison of disinfection performance of UVC-LED and conventional upper-room UVGI systems. Indoor Air 30, 180–191 (2020). doi: 10.1111/ina.12619 [21] Morrison G, Shaughnessy R, Shu S. Setting maximum emission rates from ozone emitting consumer appliances in the United States and Canada. Atmos Environ 45, 2009–2016 (2011). doi: 10.1016/j.atmosenv.2010.11.058 [22] Schalk S, Adam V, Arnold E, Brieden K, Voronov A et al. UV-lamps for disinfection and advanced oxidation-lamp types, technologies and applications. IUVA News 8, 32–37 (2006). [23] >Li DB, Jiang K, Sun XJ, Guo CL. AlGaN photonics: recent advances in materials, and ultraviolet devices. Adv Opt Photonics 10, 43–110 (2018). doi: 10.1364/AOP.10.000043 [24] Shatalov M, Sun WH, Lunev A, Hu XH, Dobrinsky A et al. AlGaN deep-ultraviolet light-emitting diodes with external quantum efficiency above 10%. Appl Phys Express 5, 082101 (2012). doi: 10.1143/APEX.5.082101 [25] Takano T, Mino T, Sakai J, Noguchi N, Tsubaki K et al. Deep-ultraviolet light-emitting diodes with external quantum efficiency higher than 20% at 275 nm achieved by improving light-extraction efficiency. Appl Phys Express 10, 031002 (2017). doi: 10.7567/APEX.10.031002 [26] Khan MA, Maeda N, Yun J, Jo M, Yamada Y et al. Achieving 9.6% efficiency in 304 nm p-AlGaN UVB LED via increasing the holes injection and light reflectance. Sci Rep 12, 2591 (2022). doi: 10.1038/s41598-022-04876-x [27] Luo W, Li T, Li YD, Wang HJ, Yuan Y et al. Watts-level ultraviolet-C LED integrated light sources for efficient surface and air sterilization. J Semicond 43, 072301 (2022). doi: 10.1088/1674-4926/43/7/072301 [28] Suzuki A, Emoto A, Shirai A, Nagamatsu K. Ultraviolet light-emitting diode (UV-LED) sterilization of citrus bacterial canker disease targeted for effective decontamination of citrus sudachi fruit. Biocontrol Sci 27, 1–7 (2022). doi: 10.4265/bio.27.1 [29] Lee YW, Yoon HD, Park JH, Ryu UC. Application of 265-nm UVC LED lighting to sterilization of typical gram negative and positive bacteria. J Korean Phys Soc 72, 1174–1178 (2018). doi: 10.3938/jkps.72.1174 [30] Kim BS, Youm S, Kim YK. Sterilization of harmful microorganisms in hydroponic cultivation using an ultraviolet LED light source. Sensor Mater 32, 3773–3785 (2020). doi: 10.18494/SAM.2020.2979 [31] Huang SY, Lin JC, Huang XQ, Wang WK. Large-area 280 nm LED flexible sterilization light source with improved thermal performance. Optik 248, 168109 (2021). doi: 10.1016/j.ijleo.2021.168109 [32] Oguma K, Rattanakul S, Masaike M. Inactivation of health-related microorganisms in water using UV light-emitting diodes. Water Supply 19, 1507–1514 (2019). doi: 10.2166/ws.2019.022 [33] Liu SF, Luo W, Li D, Yuan Y, Tong W et al. Sec-eliminating the SARS-CoV-2 by AlGaN based high power deep ultraviolet light source. Adv Funct Mater 31, 2008452 (2021). doi: 10.1002/adfm.202008452 [34] Bormann M, Alt M, Schipper L, de Sand L, Otte M et al. Disinfection of SARS-CoV-2 contaminated surfaces of personal items with UVC-LED disinfection boxes. Viruses 13, 598 (2021). doi: 10.3390/v13040598 [35] Shimoda H, Matsuda J, Iwasaki T, Hayasaka D. Efficacy of 265-nm ultraviolet light in inactivating infectious SARS-CoV-2. J Photochem Photobiol 7, 100050 (2021). doi: 10.1016/j.jpap.2021.100050 [36] Gerchman Y, Mamane H, Friedman N, Mandelboim M. UV-LED disinfection of Coronavirus: Wavelength effect. J Photochem Photobiol B 212, 112044 (2020). doi: 10.1016/j.jphotobiol.2020.112044 [37] Inagaki H, Saito A, Sugiyama H, Okabayashi T, Fujimoto S. Rapid inactivation of SARS-CoV-2 with deep-UV LED irradiation. Emerg Microbes Infect 9, 1744–1747 (2020). doi: 10.1080/22221751.2020.1796529 [38] Kojima M, Mawatari K, Emoto T, Nishisaka-Nonaka R, Bui TKN et al. Irradiation by a combination of different peak-wavelength ultraviolet-light emitting diodes enhances the inactivation of influenza A viruses. Microorganisms 8, 1014 (2020). doi: 10.3390/microorganisms8071014 [39] Sun XJ, Li DB, Chen YR, Song H, Jiang H et al. In situ observation of two-step growth of AlN on sapphire using high-temperature metal-organic chemical vapour deposition. CrystEngComm 15, 6066–6073 (2013). doi: 10.1039/c3ce40755a [40] Jiang K, Sun XJ, Ben JW, Jia YP, Liu HN et al. The defect evolution in homoepitaxial AlN layers grown by high-temperature metal-organic chemical vapor deposition. CrystEngComm 20, 2720–2728 (2018). doi: 10.1039/C8CE00287H [41] Zhang LS, Xu FJ, Wang JM, He CG, Guo WW et al. High-quality AlN epitaxy on nano-patterned sapphire substrates prepared by nano-imprint lithography. Sci Rep 6, 35934 (2016). doi: 10.1038/srep35934 [42] Banal RG, Funato M, Kawakami Y. Initial nucleation of AlN grown directly on sapphire substrates by metal-organic vapor phase epitaxy. Appl Phys Lett 92, 241905 (2008). doi: 10.1063/1.2937445 [43] Ben JW, Xun XJ, Jia YP, Jiang K, Shi ZM et al. Defect evolution in AlN templates on PVD-AlN/sapphire substrates by thermal annealing. CrystEngComm 20, 4623–4629 (2018). doi: 10.1039/C8CE00770E [44] Susilo N, Hagedorn S, Jaeger D, Miyake H, Zeimer U et al. AlGaN-based deep UV LEDs grown on sputtered and high temperature annealed AlN/sapphire. Appl Phys Lett 112, 041110 (2018). doi: 10.1063/1.5010265 [45] Uesugi K, Kuboya S, Shojiki K, Xiao SY, Nakamura T et al. 263 nm wavelength UV-C LED on face-to-face annealed sputter-deposited AlN with low screw- and mixed-type dislocation densities. Appl Phys Express 15, 055501 (2022). doi: 10.35848/1882-0786/ac66c2 [46] Miyake H, Lin CH, Tokoro K, Hiramatsu K. Preparation of high-quality AlN on sapphire by high-temperature face-to-face annealing. J Cryst Growth 456, 155–159 (2016). doi: 10.1016/j.jcrysgro.2016.08.028 [47] Xiao SY, Suzuki R, Miyake H, Harada S, Ujihara T. Improvement mechanism of sputtered AlN films by high-temperature annealing. J Cryst Growth 502, 41–44 (2018). doi: 10.1016/j.jcrysgro.2018.09.002 [48] Himwas C, Songmuang R, Dang LS, Bleuse J, Rapenne L et al. Thermal stability of the deep ultraviolet emission from AlGaN/AlN Stranski-Krastanov quantum dots. Appl Phys Lett 101, 241914 (2012). doi: 10.1063/1.4770075 [49] Himwas C, den Hertog M, Bellet-Amalric E, Songmuang R, Donatini F et al. Enhanced room-temperature mid-ultraviolet emission from AlGaN/AlN Stranski-Krastanov quantum dots. J Appl Phys 116, 023502 (2014). doi: 10.1063/1.4887140 [50] Liu SF, Yuan Y, Huang LJ, Zhang J, Wang T et al. Drive high power UVC-LED wafer into low-cost 4-inch era: effect of strain modulation. Adv Funct Mater 32, 2112111 (2022). doi: 10.1002/adfm.202112111 [51] Jiang K, Sun XJ, Shi ZM, Zang H, Ben JW et al. Quantum engineering of non-equilibrium efficient p-doping in ultra-wide band-gap nitrides. Light Sci Appl 10, 69 (2021). doi: 10.1038/s41377-021-00503-y [52] Han QL, Chang C, Li L, Klenk C, Cheng JK et al. Sumoylation of influenza A virus nucleoprotein is essential for intracellular trafficking and virus growth. J Virol 88, 9379–9390 (2014). doi: 10.1128/JVI.00509-14 [53] Xiong HL, Wu YT, Cao JL, Yang R, Liu YX et al. Robust neutralization assay based on SARS-CoV-2 S-protein-bearing vesicular stomatitis virus (VSV) pseudovirus and ACE2-overexpressing BHK21 cells. Emerg Microbes Infect 9, 2105–2113 (2020). doi: 10.1080/22221751.2020.1815589 [54] Wen W, Chen C, Tang JK, Wang CY, Zhou MY et al. Efficacy and safety of three new oral antiviral treatment (molnupiravir, fluvoxamine and Paxlovid) for COVID-19: a meta-analysis. Ann Med 54, 516–523 (2022). doi: 10.1080/07853890.2022.2034936 [55] Matrosovich M, Matrosovich T, Garten W, Klenk HD. New low-viscosity overlay medium for viral plaque assays. Virol J 3, 63 (2006). doi: 10.1186/1743-422X-3-63 [56] Zamora JLR, Ortega V, Johnston GP, Li J, André NM et al. Third helical domain of the Nipah virus fusion glycoprotein modulates both early and late steps in the membrane fusion cascade. J Virol 94, e00644–20 (2020). doi: 10.1128/jvi.00644-20 [57] Heying B, Tarsa EJ, Elsass CR, Fini P, DenBaars SP et al. Dislocation mediated surface morphology of GaN. J Appl Phys 85, 6470–6476 (1999). doi: 10.1063/1.370150 [58] Peng LY, Zhao DG, Liang F, Wang WJ, Liu ZS et al. Influences of gallium and nitrogen partial pressure on step-bunching and step-meandering morphology of InGaN quantum barrier layer. Mater Today Commun 29, 102923 (2021). doi: 10.1016/j.mtcomm.2021.102923 [59] Hamachi T, Tohei T, Hayashi Y, Imanishi M, Usami S et al. Propagation of threading dislocations and effects of Burgers vectors in HVPE-grown GaN bulk crystals on Na-flux-grown GaN substrates. J Appl Phys 129, 225701 (2021). doi: 10.1063/5.0053766 [60] Dong P, Yan JC, Zhang Y, Wang JX, Zeng JP et al. AlGaN-based deep ultraviolet light-emitting diodes grown on nano-patterned sapphire substrates with significant improvement in internal quantum efficiency. J Cryst Growth 395, 9–13 (2014). doi: 10.1016/j.jcrysgro.2014.02.039 [61] He CG, Zhao W, Wu HL, Zhang S, Zhang K et al. High-quality AlN film grown on sputtered AlN/sapphire via growth-mode modification. Cryst Growth Des 18, 6816–6823 (2018). doi: 10.1021/acs.cgd.8b01045 [62] Liu B, Zhang R, Zheng JG, Ji XL, Fu DY et al. Composition pulling effect and strain relief mechanism in AlGaN/AlN distributed Bragg reflectors. Appl Phys Lett 98, 261916 (2011). doi: 10.1063/1.3605681 [63] Jiang K, Sun XJ, Ben JW, Shi ZM, Jia YP et al. Suppressing the compositional non-uniformity of AlGaN grown on a HVPE-AlN template with large macro-steps. CrystEngComm 21, 4864–4873 (2019). doi: 10.1039/C9CE00608G [64] LaBarre DD, Lowy RJ. Improvements in methods for calculating virus titer estimates from TCID50 and plaque assays. J Virol Methods 96, 107–126 (2001). doi: 10.1016/S0166-0934(01)00316-0 [65] Hirose R, Itoh Y, Ikegaya H, Miyazaki H, Watanabe N et al. Differences in environmental stability among SARS-CoV-2 variants of concern: both omicron BA. 1 and BA. 2 have higher stability. Clin Microbiol Infect 28, 1486–1491 (2022). doi: 10.1016/j.cmi.2022.05.020 -
Supplementary Information
Supplementary information for Rapid inactivation of human respiratory RNA viruses by deep ultraviolet irradiation from light-emitting diodes on a high-temperature-annealed AlN/Sapphire template 
-
Access History
Article Metrics
-
Figure 1.
Cross-sectional BFDB STEM images (<1-100>) of the n-AlGaN epilayer grown on a HTA AlN/Sapphire template. (a, b) and (c, d) are the images without and with SLs, respectively. (a, c) and (b, d) are taken with g = (0002) and (11-20), respectively. For one sample, the images with different g vectors are taken in the same area. The white arrows in (b) and (d) denote the edge dislocations in the HTA AlN/Sapphire template.
-
Figure 2.
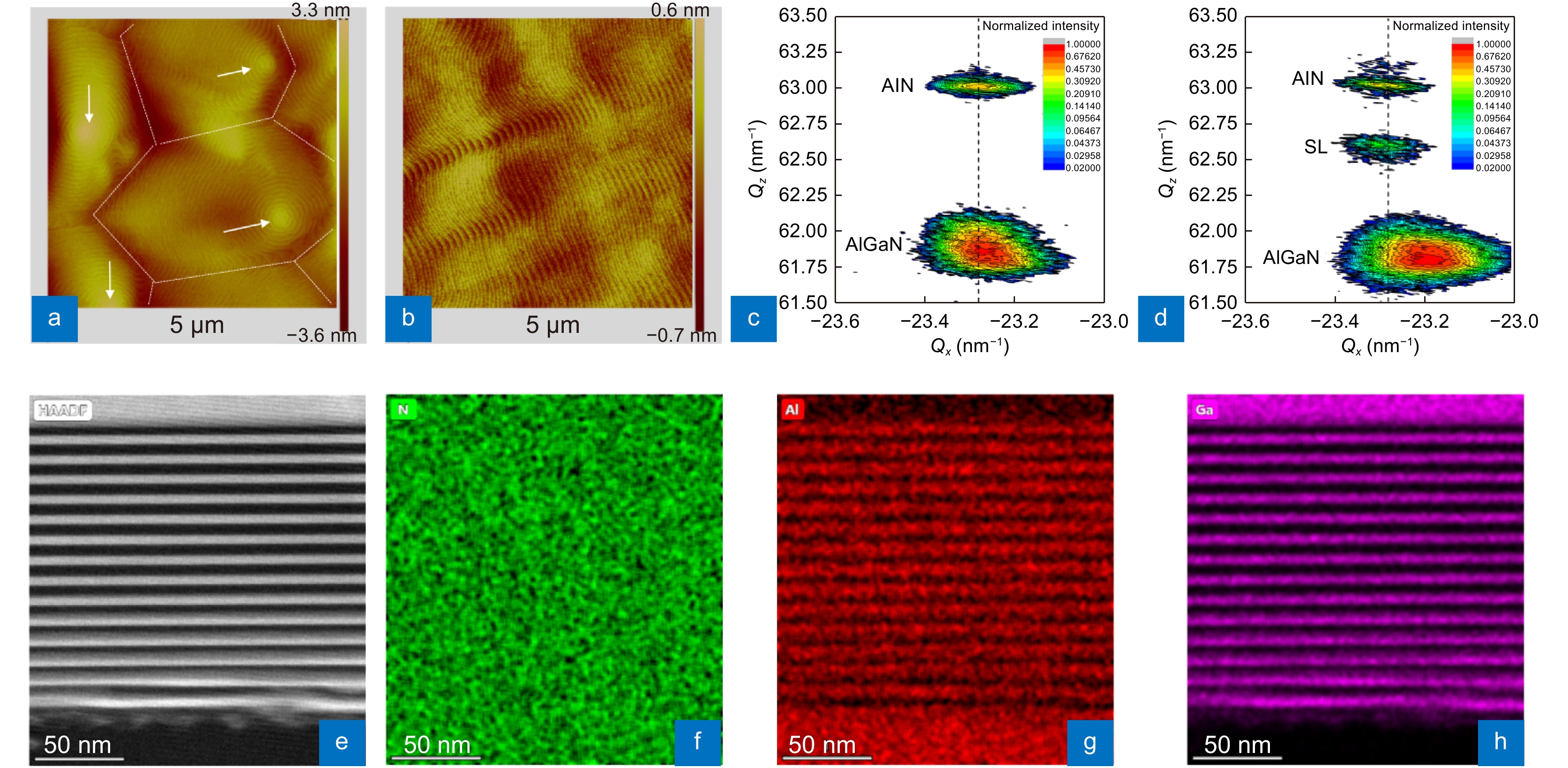
Surface and strain states of the n-AlGaN epilayer grown on a HTA AlN/Sapphire template and structure of the SLs. (a, b) and (c, d) are AFM images (5 μm × 5 μm) and XRD RSMs of the (–105) planes for wafers without/with SLs. (e–h) Cross-sectional HAADF STEM image (<11–20>) of an AlN/AlGaN SLs area and corresponding EDS mappings of N, Al, and Ga elements.
-
Figure 3.
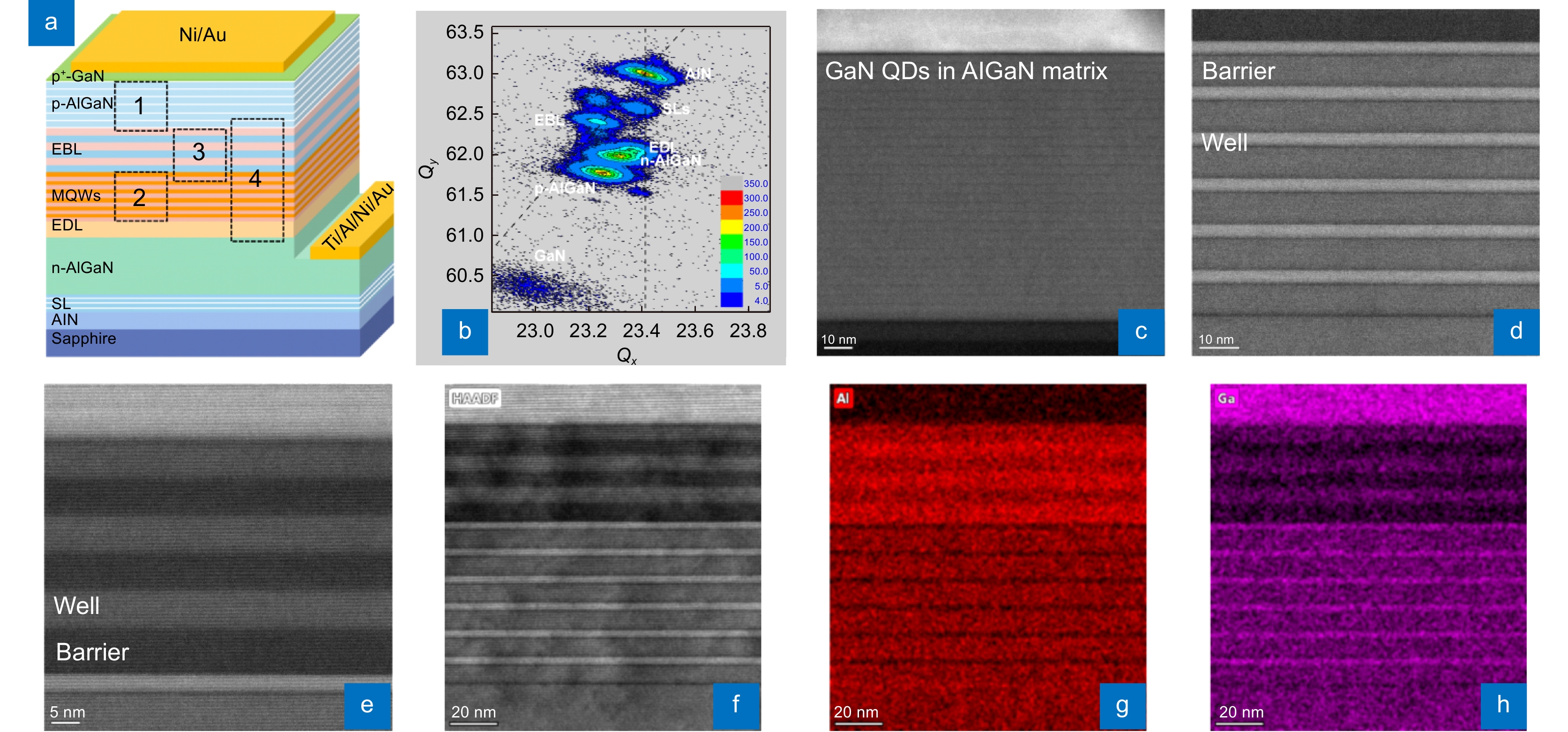
DUV LED structure grown on a HTA AlN/Sapphire template. (a) Structure diagram of the DUV LED. (b) XRD RSM of the (105) plane of the DUV LED wafer. (c–e) Cross-sectional HAADF STEM images (<11–20>) of the p-AlGaN HIL, MQWs, and EBL (areas 1, 2, and 3 in Fig. 3(a)) of the DUV LED, respectively. (f–h) HAADF STEM image (<11–20>) of the DUV LED (area 4 in Fig. 3(a)) and corresponding EDS mappings of Al and Ga.
-
Figure 4.
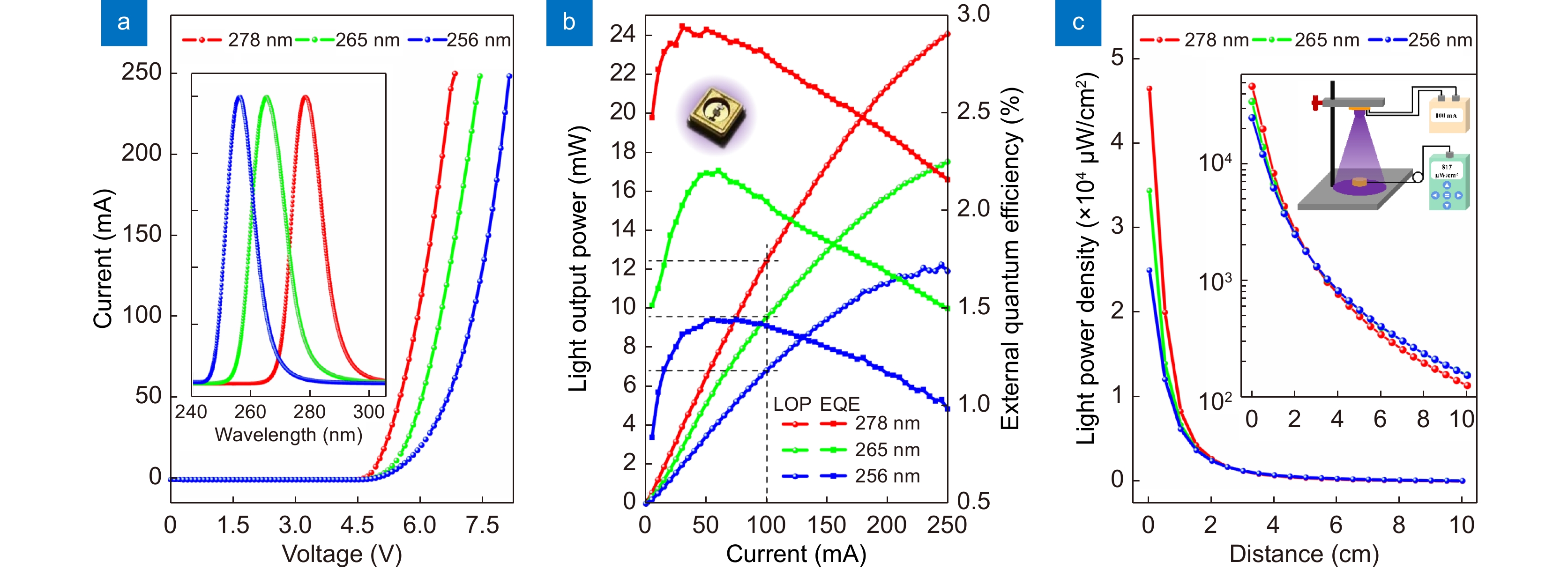
Performance characterizations of the three DUV LEDs. (a) IV curves. The inset shows the normalized EL spectra at the current of 100 mA. (b) LOP and EQE curves. The inset shows a picture of a typical flip-chip packaged LED. (c) Distance-dependent LPD at the current of 100 mA. The insets show the log-scale graph and measurement geometry.
-
Figure 5.
Inactivation effects of the DUV LEDs on IAV and SARS-CoV-2. (a) Inactivation efficiency for IAV with an initial titer of 2.3×104 PFUs in 60 μL. The inset shows the results for a higher initial titer of 2.3×105 PFUs in 60 μL. (b) Irradiation time-dependent viral inactivation effects for IAV from 2 to 12 cm. The inset shows the live viral titer after 10 s at 8 and 12 cm. (c) Viral inactivation effects of the 256 nm-LED for IAV in 10 s at 4 cm. The viral titers determined by the ratio of PFUs (bar chart) and log10 reduction (cyan line) are shown. (d) Live viral titer after 10 s irradiation for IAV at 4 cm on different materials. Values are presented as the means±SDs (n=3, n=number of independent replicates). (e) Inactivation efficiency with an initial titer of 2.3×104 PFUs in 60 μL for SARS-CoV-2. Values are presented as the means±SDs (n=2, n=number of biological replicates). (f) Inactivation effects of the 256 nm-LED for different pSARS-CoV-2 variants. BHK21-hACE2 cells are infected for 24 h with pseudo-SARS-CoV-2 irradiated or not, and then, the luciferase activity is measured to reflect the virus entry efficiency. Time-dependent effects at 4 cm are evaluated by measuring the luciferase activity for individual pSARS-CoV-2. The dotted line represents the detection limit. Values are presented as the means±SDs (n=3, n=number of independent replicates). **p<0.01.
-
Figure 6.
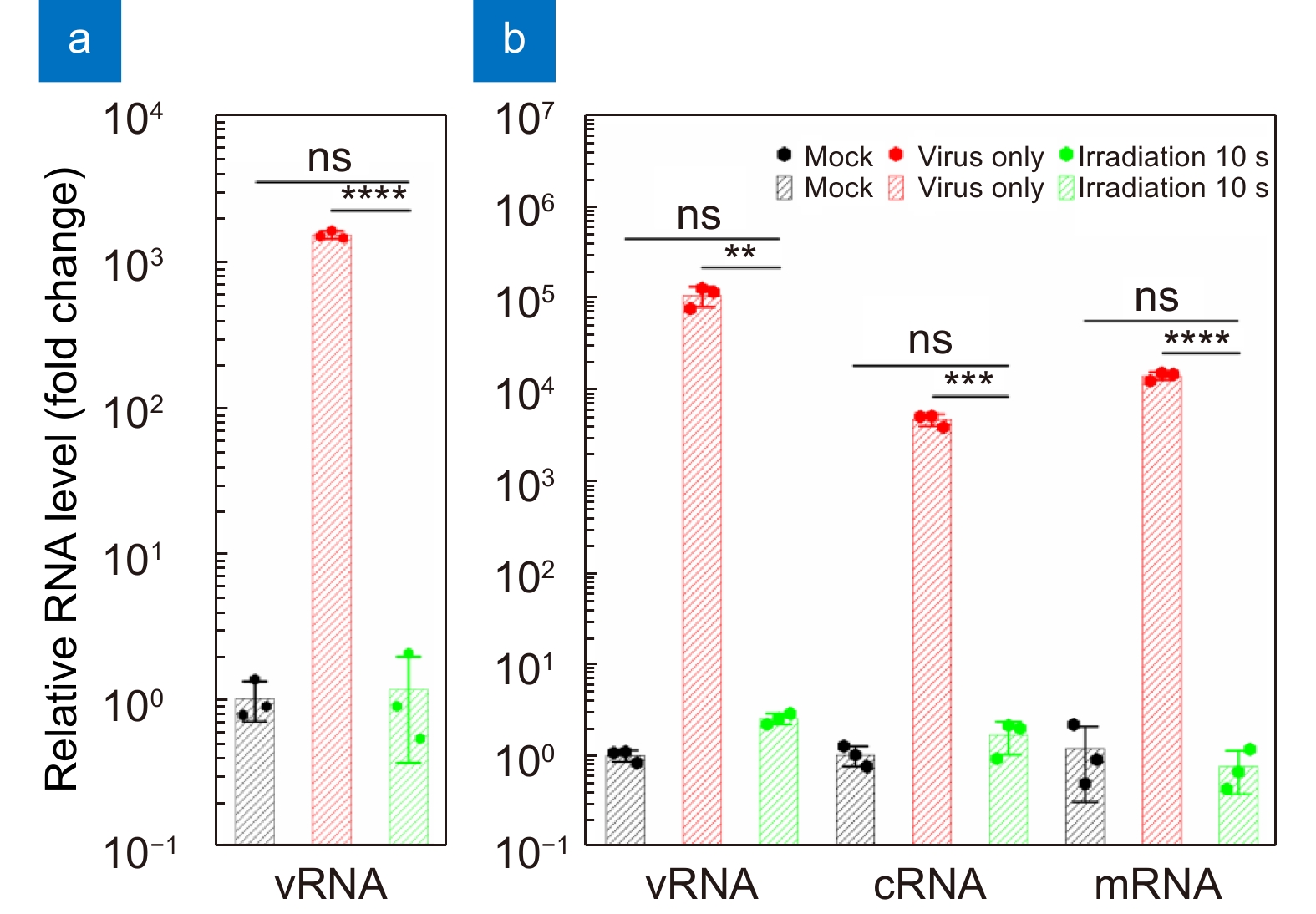
Total relative RNA levels in the cell (a) supernatants and (b) lysates infected by a virus suspension irradiated for 10 s at 4 cm or not after 24 h. The relative RNA level is normalized to that of the mock (non-infected) group. Values are presented as the means±SDs (n=3, n=number of independent replicates). **p < 0.01, ***p < 0.001, ****p < 0.0001.

 E-mail Alert
E-mail Alert RSS
RSS


 DownLoad:
DownLoad: